The ability to create clots to limit blood loss from sites of vascular injury is balanced by systems designed to limit clot formation and to dissolve existing clots. The blood contains natural anticoagulants that act continuously to inhibit clot formation. Fibrinolytic agents dissolve existing clots. In doing so, blood flow through the vascular system can be maintained.
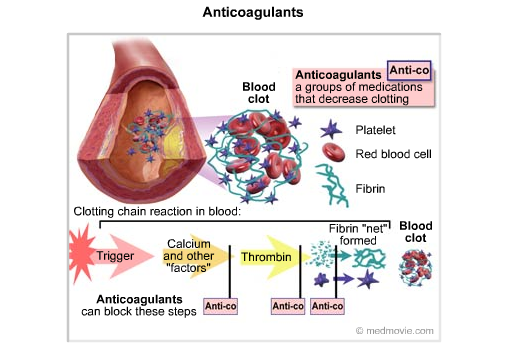
Anticoagulants reduce the ability of clots to form and also prevent unrestricted clot growth. Clinically, heparin, warfarin, and aspirin are the most commonly used anticoagulants. Heparin acts immediately to potentiate antithrombin III action and prevent clot formation. Warfarin interferes with the vitamin K-dependent hepatic synthesis of clotting factors. Consequently, treatment with warfarin has little immediate effect but is useful for chronic suppression of clotting ability. Aspirin is classified as an anticoagulant, but it acts on platelets to reduce their clotting ability by blocking thromboxane production rather than acting on the clotting cascade. In vitro clot formation can be prevented by the Ca++ chelators citrate, EGTA, and EDTA. Protein C, which is activated by the clotting factor thrombin, can prevent further clot growth, and thus the normal clotting cascade produces an enzyme that helps stop the clotting process.
Fibrinolysis is the dissolution of clots, primarily due to action of the enzyme plasmin. The fibrinolytic mechanism is activated a few hours after clot formation. Plasmin is formed by the action of tissue plasminogen activator on the plasma protein plasminogen. Plasmin is a proteolytic enzyme that dissolves fibrin, fibrinogen, and factors V and VIII. Fibrin degradation products produced by clot lysis also act as anticoagulants. Plasmin additionally is formed from plasminogen by streptokinase, staphylokinase, and urokinase as well as intrinsic pathway components. Clinical trials contrasting the effectiveness of the different fibrinolytic agents in myocardial infarction are under way.
Disorders can occur due to insufficient or inappropriate clotting activity. Insufficient clotting activity can be due to thrombocytopenia, the lack of thrombocytes due to impaired production, enhanced breakdown, or enhanced use of thrombocytes. Thrombocytopenia leads to a prolonged bleeding time. In contrast, thrombocytosis is defined as a platelet count greater than 800,000, but it also is characterized by inappropriate bleeding.
Inappropriate clotting can cause thrombosis, the occurrence of a clot within a blood vessel. This can occur following endothelial damage, blood stasis, or enhanced coagulability. An embolus is a clot that breaks free of its attachment site and is carried within the circulation. Emboli are trapped by the branching pattern characteristic of the precapillary side of the circulation. Venous emboli are trapped in the lungs, which have enhanced clot lysis abilities. Left ventricular emboli become trapped in the systemic circulation and block blood flow to the areas distal to the occlusion. Emboli are often responsible for strokes and myocardial infarction.
Blood vessels break and are repaired continuously in the body. The multiple and complex interactions between clot formation, clot lysis, and anticoagulation allow normal vessel repair without precipitation of a massive clot throughout the vascular system. Functional tests of coagulation include bleeding time, clot retraction, and platelet aggregation.
Laboratory Values for Intrinsic Pathway Activation
| Hematologic | ||
| Parameter | Typical Value | Reference Range |
| Bleeding time | 5 min | 2-7 min |
| Erythrocyte count Male Female | 5.0 million/mm3 4.2 million/mm3 | 4.3-5.9 million/mm3 3.5-5.5 million/mm3 |
| Hematocrit Male Female | 48% 42% | 41-53% 36-46% |
| Hemoglobin, total Male Female | 15 g/dL 14 g/dL | 13.5-17.5 g/dL 12.0-16.0 g/dL |
| Partial thromboplastin time (activated) | 35 sec | 28-40 sec |
| Platelet count | 300,000 mm3 | 150,000-400,000 mm3 |
| Prothrombin time | 13 sec | 11-15 sec |
| Reticulocyte count | 1% | 0.5-1.5% of red cells |
| Thrombin time | <2 sec deviation from control |

 (63 votes, average: 4.46 out of 5)
(63 votes, average: 4.46 out of 5)