Blood flow serves multiple functions. Matching of blood flow to metabolic needs is complex in organs that have variable metabolic rates, such as skeletal muscle. Local regulation of blood flow is well developed in tissues that have a low tolerance for ischemia, such as the brain and the heart. Some regional circulations serve functions other than tissue nutrition. For example, the cutaneous circulation assists in the regulation of body temperature, the renal circulation transports waste products to the kidneys for elimination, the splanchnic circulation transports absorbed intestinal nutrients, and the pulmonary circulation assists gas exchange. The unique cardiovascular characteristics of tissues are tied tightly to tissue function.
There is extensive sympathetic control of cutaneous vascular smooth muscle and therefore cutaneous blood flow. A decrease in body core temperature leads to vasoconstriction, which decreases the radiant loss of heat. Conversely, an increased body core temperature dilates the arteriovenous anastomoses, increasing cutaneous blood flow, and thereby increasing the radiant loss of heat.
Sympathetic cholinergic nerves innervate cutaneous sweat glands. Activation of these nerves facilitates heat loss through evaporation and indirectly dilates cutaneous vessels via bradykinin release from the sweat glands. CNS output regulates cutaneous vascular smooth muscle in vessels of the head, neck, and shoulders and can cause blushing. A countercurrent heat exchange mechanism in the arms and legs assists thermoregulation. Cool (venous) blood from extremities is warmed as it returns to the body core, and warm (arterial) blood from the body core is cooled as it flows to the extremities. In this way, blood flow to cold extremities, such as feet, hands, and ears, can be maintained to provide adequate delivery of nutrition without compromising temperature regulation.
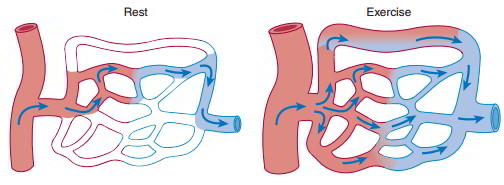
Skeletal muscle blood flow is proportionate to metabolic activity. Only 20% of skeletal muscle capillaries are perfused at rest. Skeletal muscle blood flow can increase tenfold during exercise. In resting skeletal muscle, sympathetic adrenergic nerves constrict the vasculature and reduce blood flow. Local metabolic control normally is the most powerful, and local vasodilation can override neural regulation during exercise.
Cerebral circulation provides a constant blood supply to the CNS. Multiple inflows from paired internal carotid arteries, vertebral arteries, and spinal arteries join to form the circle of Willis. The CNS is enclosed in a rigid cranium, so inflow must equal outflow or pressure may increase and damage the CNS tissue. CNS tissue is sensitive to blood flow interruption. After 5 seconds of interruption, a person becomes unconscious, and after 5 minutes, irreversible tissue damage occurs.
Cerebral blood flow regulation is primarily local, so blood flow in discrete brain areas is proportionate to metabolism. CNS autoregulation of blood flow is well developed. The CNS ischemic response is the most powerful activator of sympathetic nerves, occurring when blood pressure falls below 60 mm Hg or increased intracranial pressure prevents blood entry into cranium, called the Cushing reflex.
Splanchnic blood vessels are arranged partly in series, since the GI, spleen, and pancreas capillary beds empty into the hepatic portal vein. Intestinal blood flow is regulated by the SNS. This allows shunting of blood during the fight or flight response. A small amount of local control is seen. Intestinal blood flow also is directly influenced by GI hormones, such as vasoactive intestinal polypeptide (VIP).
Unique blood flow pathways transport intestinal blood to the liver sinusoids before the blood enters the general circulation. Normally 25% of the cardiac output goes to the liver in two vessels. The hepatic portal vein carries three quarters of the blood entering the liver, and the hepatic artery carries the remaining one quarter. The hepatic artery provides most of the O2 consumed by the liver. Both the hepatic artery and portal vein empty into a hepatic acinus. Blood flows outward from the acinus to sinusoids to hepatic veins.
The hepatic circulation has a low blood pressure. Ascites results from increased liver sinus capillary pressure, usually secondary to increased central venous pressure. The liver is an important capacitance organ and has 15% of total blood volume, half of which can be expelled under stress.
The spleen also has sinuses rather than true capillaries. The spleen stores red blood cells, and it filters and destroys fragile red blood cells. As red blood cells age, they become more rigid. Passage through the spleen deforms the red blood cells and ruptures those that are rigid.
The coronary blood flow is tightly coupled to the workload of the cardiac muscle. Cardiac muscle is almost exclusively aerobic. O2 extraction by cardiac tissue is high, so increased O2 delivery is accomplished only by increasing blood flow. Consequently, the myocardium is susceptible to damage if blood flow is interrupted. Autoregulation of coronary blood flow is well developed, with adenosine playing a major role as a local vasodilator metabolite.
Tension developed by the contracting muscle impedes coronary blood flow, particularly to the left ventricular endocardium, during systole. Consequently, left ventricular blood flow is highest during diastole. The right ventricle develops lower pressures and is perfused during ventricular systole. Blood flow to the left ventricle can be compromised by the decreased diastolic duration characteristic of rapid heart rates.
Cardiac vascular smooth muscle is innervated by sympathetic nerves. The major consequence of increased sympathetic activity to the heart, however, is an increase in myocardial blood flow. Increased sympathetic nerve activity increases cardiac work, increases heart rate, and increases contractility. The increased workload on the myocardium causes an adenosine-mediated vasodilation that overwhelms the slight tendency of sympathetics to constrict the myocardial vasculature.
Pulmonary circulation is characterized by low resistance to blood flow, and so pressures are much lower than in the systemic circulation. Pulmonary capillaries are arranged between alveoli so that blood flows in sheets. Pulmonary circulation functions include gas exchange, filtration of clots and other particulate matter, and enzyme activity, notably angiotensin I converting enzyme.
Blood entering the pulmonary circulation is O2 depleted and resembles systemic venous blood. In contrast, the tracheobronchial tree is nourished by bronchial vessels of the systemic circulation, carrying oxygenated blood. Hypoxia constricts arterioles in the lung, in contrast to the hypoxic vasodilation response in peripheral arterioles. This plays a significant role in matching pulmonary ventilation and perfusion